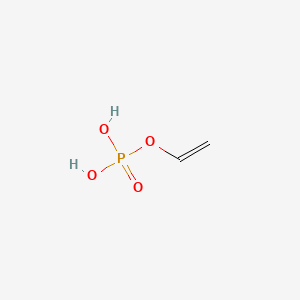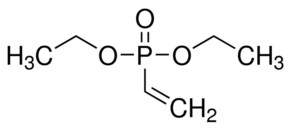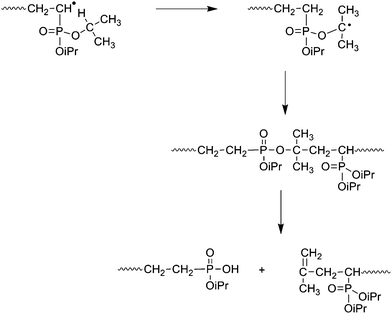
Recent progress on phosphonate vinyl monomers and polymers therefore obtained by radical (co)polymerization - Polymer Chemistry (RSC Publishing) DOI:10.1039/C1PY00276G
siRNA carrying an (E)-vinylphosphonate moiety at the 5 end of the guide strand augments gene silencing by enhanced binding to hu

Enantioselective Synthesis of γ-Phenyl-γ-amino Vinyl Phosphonates and Sulfones and Their Application to the Synthesis of Novel Highly Potent Antimalarials | ACS Omega
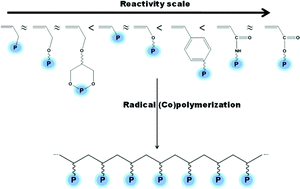
Recent progress on phosphonate vinyl monomers and polymers therefore obtained by radical (co)polymerization - Polymer Chemistry (RSC Publishing)
![An efficient deprotection method for 5′-[O,O-bis(pivaloyloxymethyl)]-(E)- vinylphosphonate containing oligonucleotides - ScienceDirect An efficient deprotection method for 5′-[O,O-bis(pivaloyloxymethyl)]-(E)- vinylphosphonate containing oligonucleotides - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0040402018310639-fx1.jpg)
An efficient deprotection method for 5′-[O,O-bis(pivaloyloxymethyl)]-(E)- vinylphosphonate containing oligonucleotides - ScienceDirect

Molecules | Free Full-Text | S-Acyl-2-Thioethyl: A Convenient Base-Labile Protecting Group for the Synthesis of siRNAs Containing 5′-Vinylphosphonate
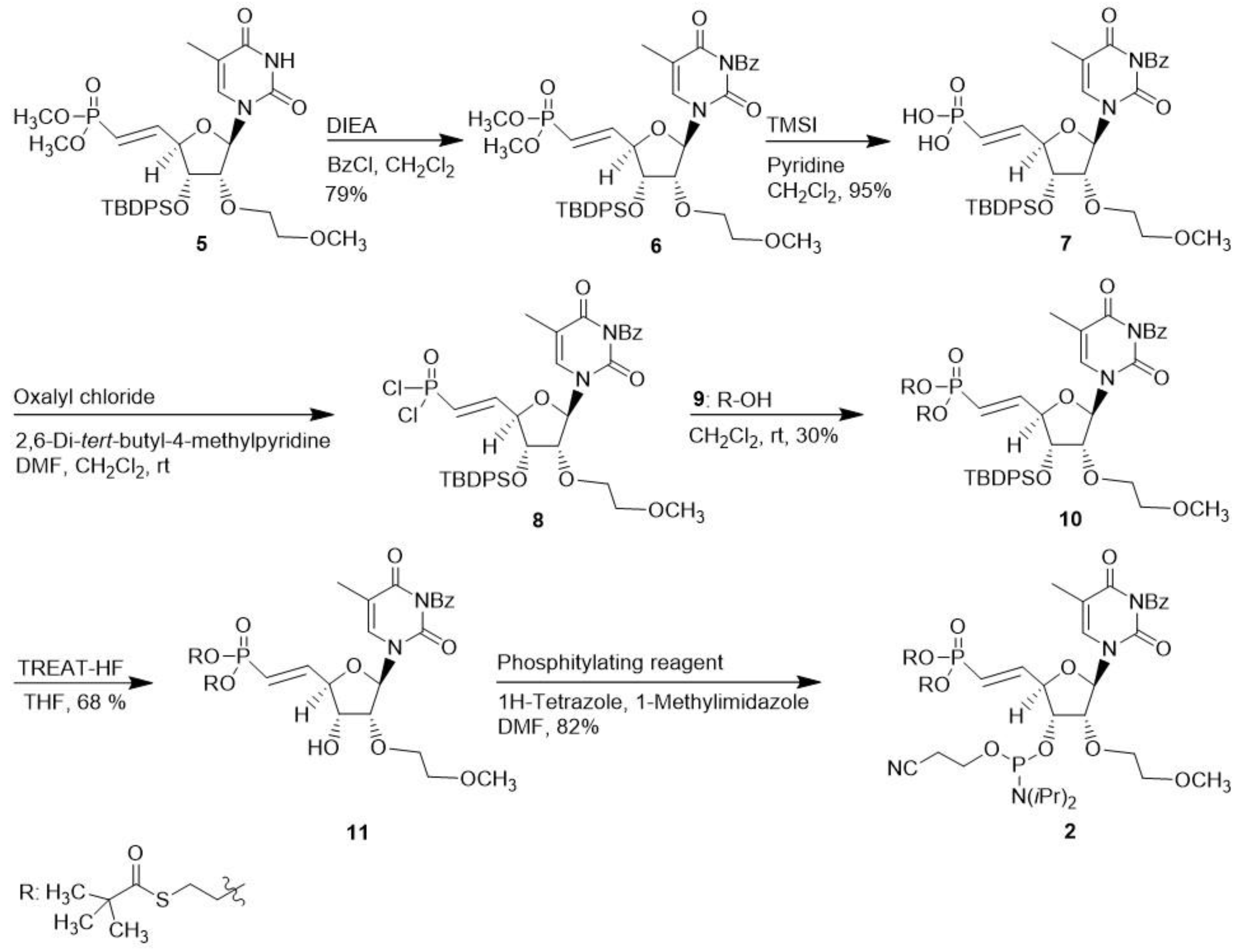
Molecules | Free Full-Text | S-Acyl-2-Thioethyl: A Convenient Base-Labile Protecting Group for the Synthesis of siRNAs Containing 5′-Vinylphosphonate

5′‐(E)‐Vinylphosphonate: A Stable Phosphate Mimic Can Improve the RNAi Activity of siRNA–GalNAc Conjugates - Parmar - 2016 - ChemBioChem - Wiley Online Library
![PDF] 5΄-Vinylphosphonate improves tissue accumulation and efficacy of conjugated siRNAs in vivo | Semantic Scholar PDF] 5΄-Vinylphosphonate improves tissue accumulation and efficacy of conjugated siRNAs in vivo | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2b867e231dd779e9564407294be43a794b82b898/5-Figure2-1.png)
PDF] 5΄-Vinylphosphonate improves tissue accumulation and efficacy of conjugated siRNAs in vivo | Semantic Scholar
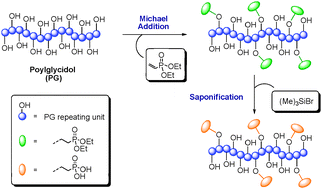
Post-polymerization functionalization of linear polyglycidol with diethyl vinylphosphonate - Chemical Communications (RSC Publishing)
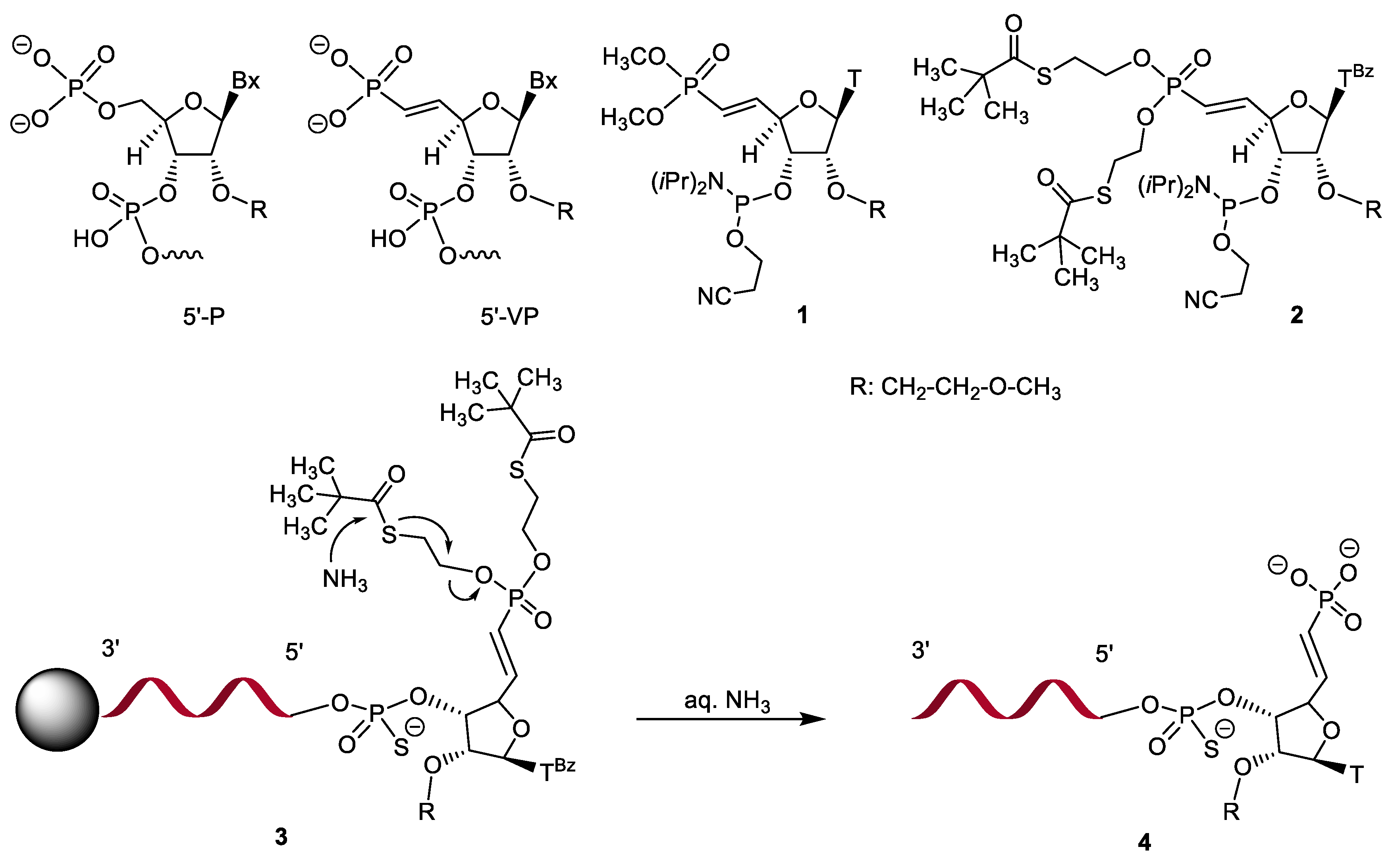
Molecules | Free Full-Text | S-Acyl-2-Thioethyl: A Convenient Base-Labile Protecting Group for the Synthesis of siRNAs Containing 5′-Vinylphosphonate