Hylenex Package: Hylenex Hyaluronidase, Human Recombinant, Preservative Free 150 Unit / mL Injection Single Dose Vial Box of 4 x 1 mL - Merit Pharmaceutical

Hyaluronidase Injection IP at Rs 215.65/1pc | Lotus Medi Sciences Pvt Ltd | Nagpur | ID: 21121541662
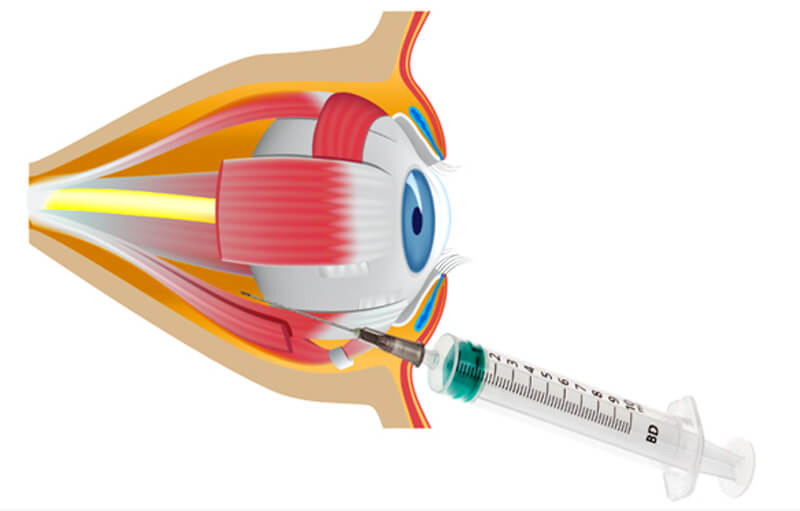
Is there a role for retrobulbar hyaluronidase in hyaluronic acid vascular embolism related vision loss? | The PMFA Journal
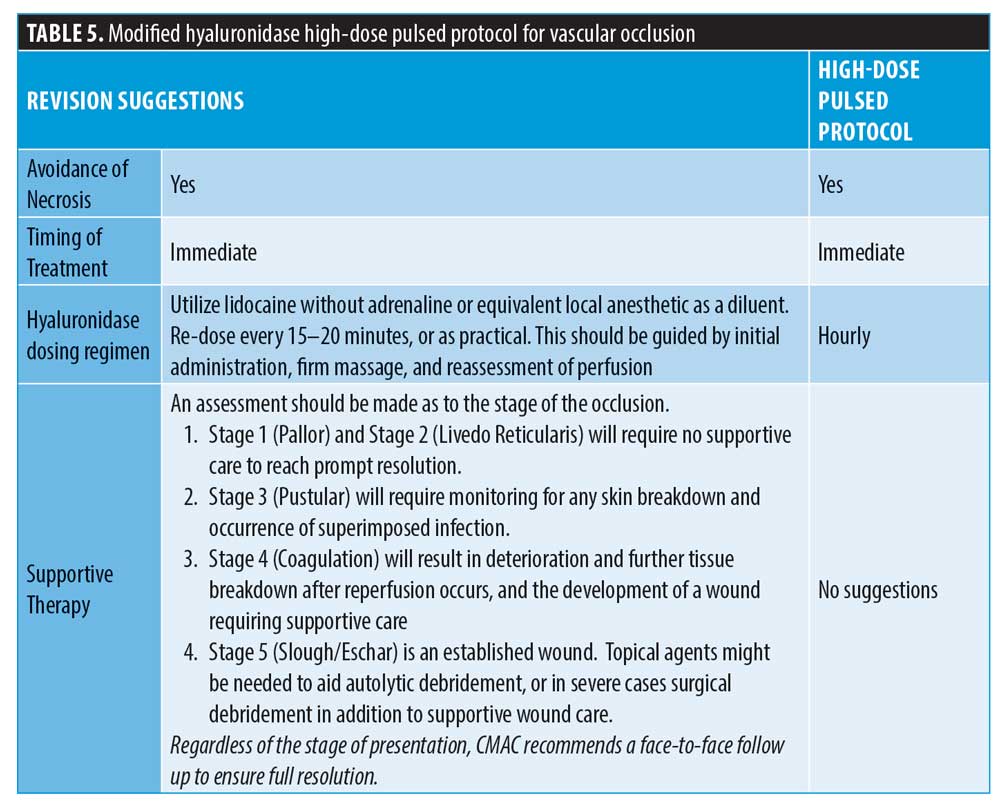
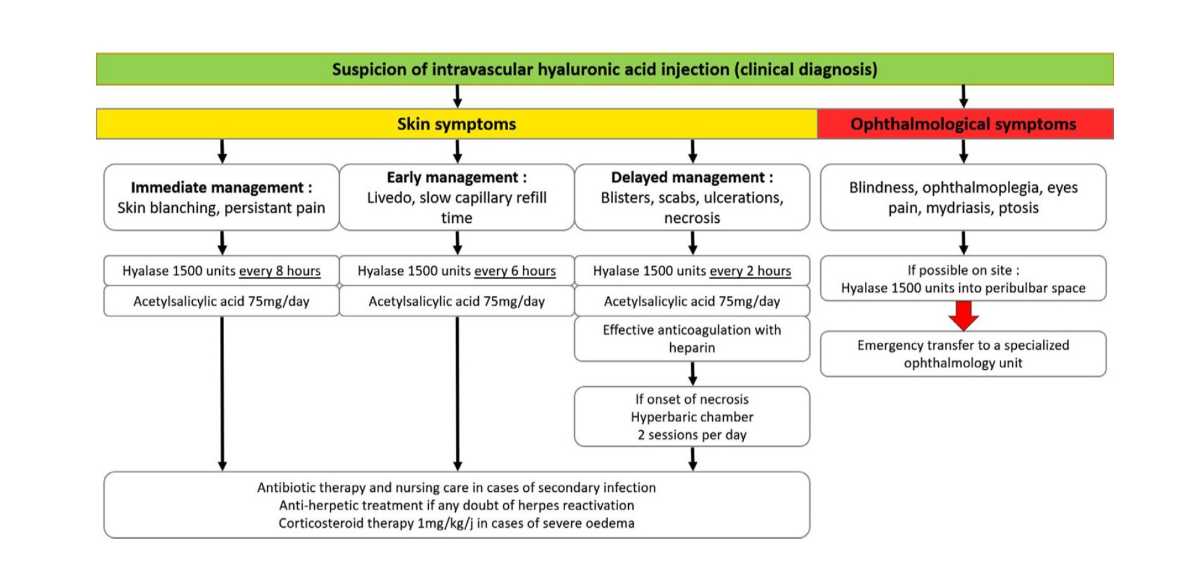
Guideline for the Safe Use of Hyaluronidase in Aesthetic Medicine, Including Modified High-dose Protocol – JCAD | The Journal of Clinical and Aesthetic Dermatology

Guideline for the Safe Use of Hyaluronidase in Aesthetic Medicine, Including Modified High-dose Protocol – JCAD | The Journal of Clinical and Aesthetic Dermatology

Degradation of Hylauronic Acid Fillers Using Hyaluronidase in an In Vivo Model - JDDonline - Journal of Drugs in Dermatology
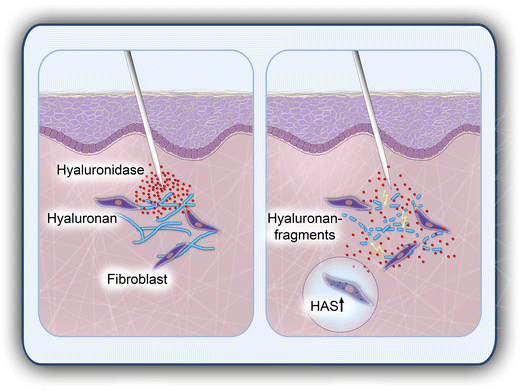
Hyaluronidase: from clinical applications to molecular and cellular mechanisms | European Journal of Medical Research | Full Text

Table 1 from The role of hyaluronidase in the treatment of complications from hyaluronic acid dermal fillers. | Semantic Scholar

Amphadase® Dispersion Agent Hyaluronidase, Bovine 150 Unit / mL Subcutaneous Injection Single Dose Vial 1 Ml - Suprememed
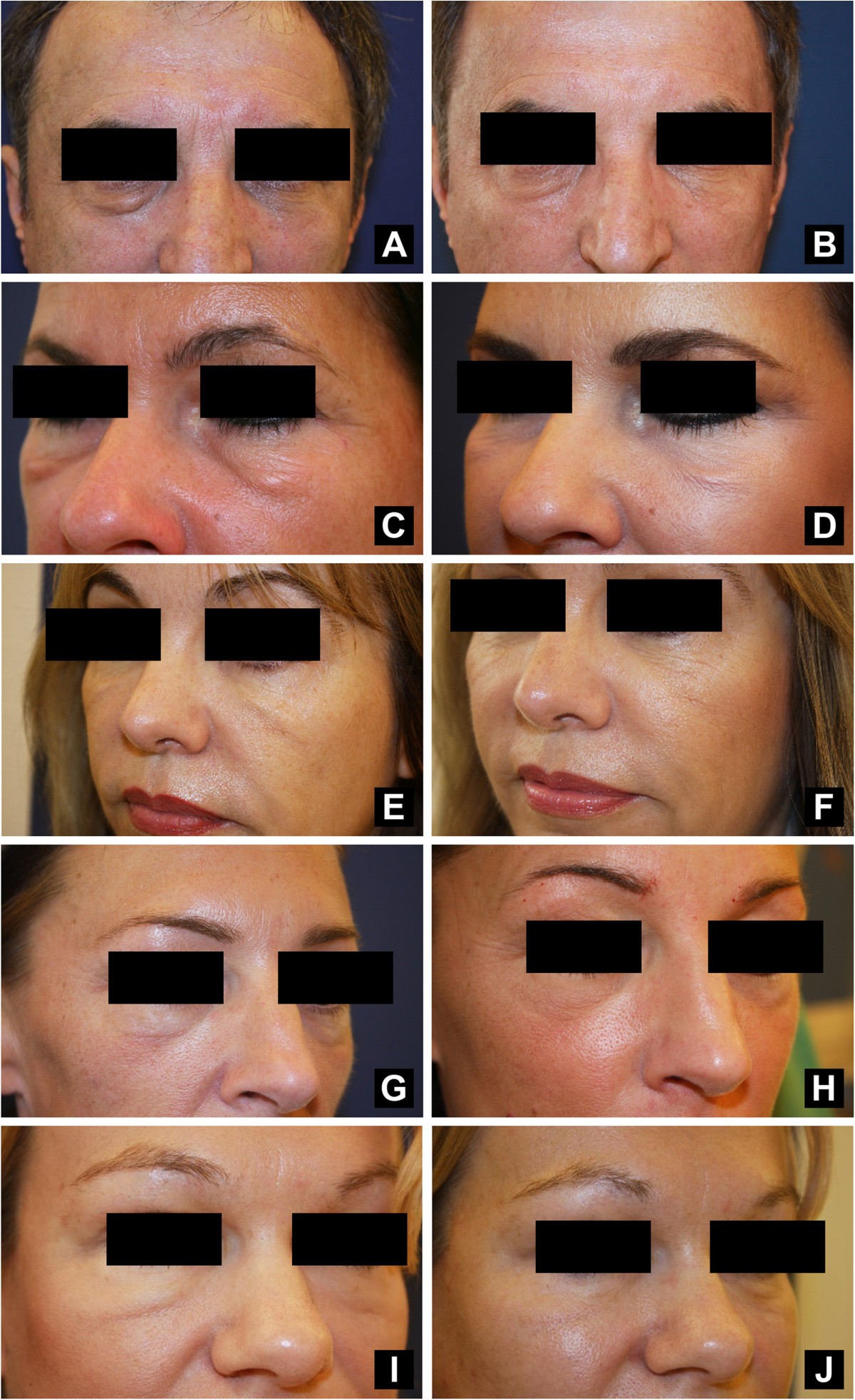
Hyaluronidase injection for the treatment of eyelid edema: a retrospective analysis of 20 patients | European Journal of Medical Research | Full Text