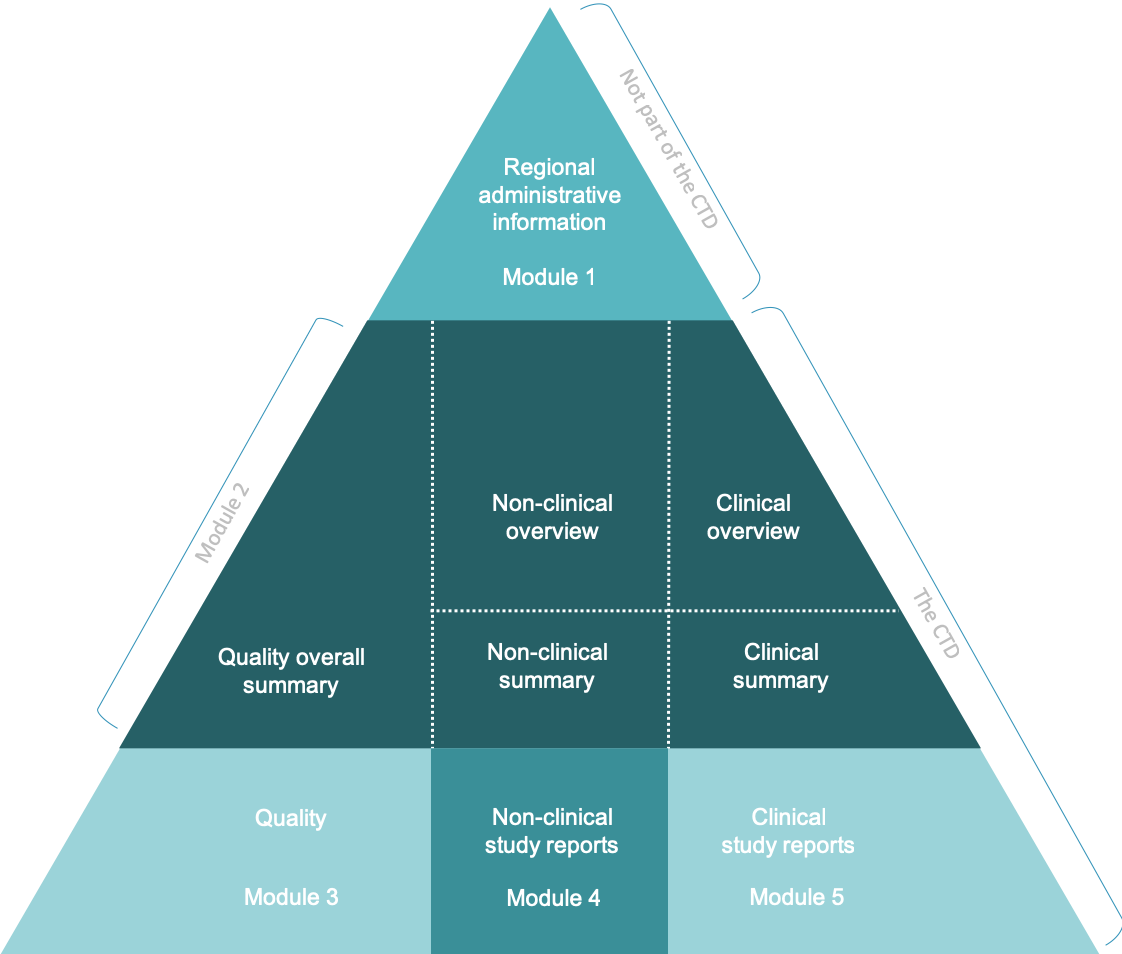
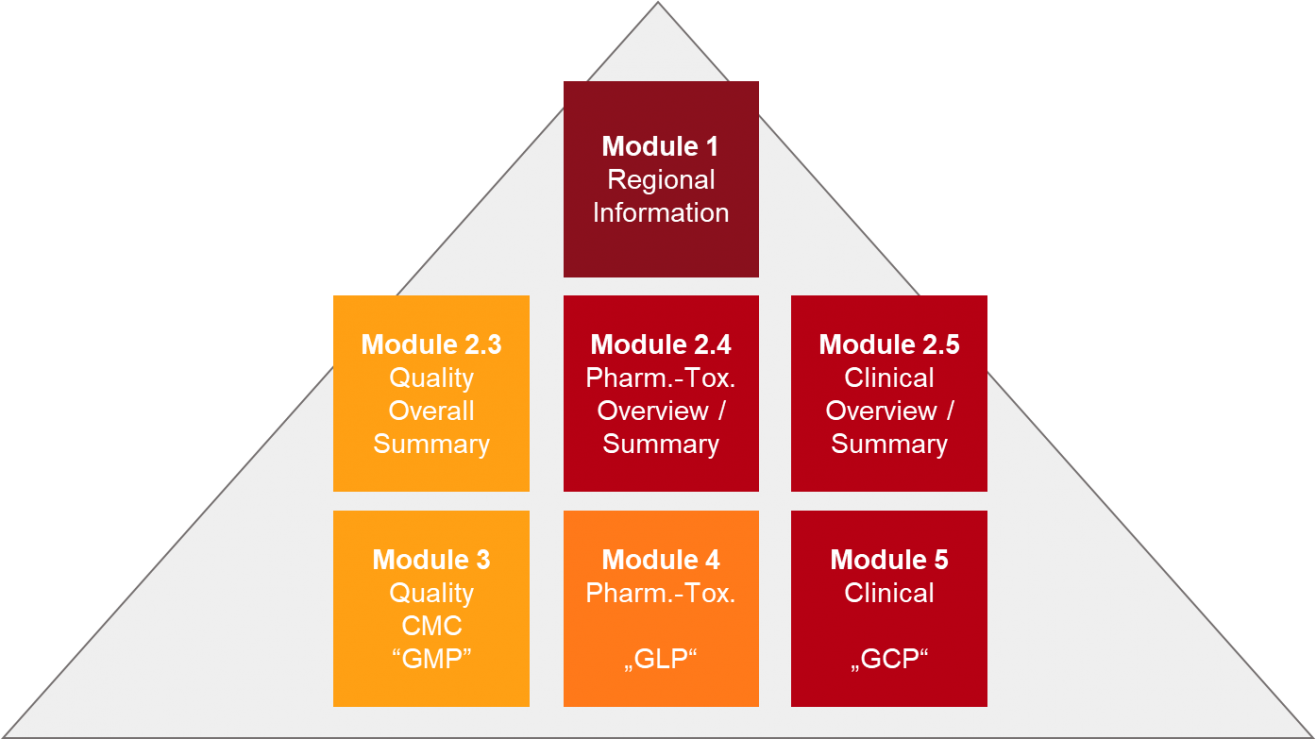
The Organisation for Professionals in Regulatory Affairs - TOPRA - Now available ONLINE How to effectively prepare and manage module 3 of the dossier covering pharmaceutical (CMC) information. Get up to date

The Organisation for Professionals in Regulatory Affairs - TOPRA - How to effectively prepare and manage module 3 of the dossier covering pharmaceutical (CMC) information. Get up to date with the latest

The Future of CMC Regulatory Submissions: Streamlining Activities Using Structured Content and Data Management - ScienceDirect

Transitioning Chemistry, Manufacturing, and Controls Content With a Structured Data Management Solution: Streamlining Regulatory Submissions - Journal of Pharmaceutical Sciences

PDF) Preparation and Review of Chemistry, Manufacturing and Control (CMC) Sections of CTD Dossier for Marketing Authorization | Dhruvi Patel - Academia.edu

Regulatory Affairs Explained Series Episode 5 | Module 3 - Chemistry, Manufacturing & Controls (CMC) - YouTube

ICH CTD QUALITY Part -CMC Module 3 Drug Substance Video by Rajashri Ojha at Raaj PharmaeLearning - YouTube
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan