Prevalence of side-effects associated with the booster dose of Pfizer- BioNTech (BNT162b2) of COVID-19 Vaccine among vaccinated adults in the Eastern province of Saudi Arabia - ScienceDirect

Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study - The Lancet
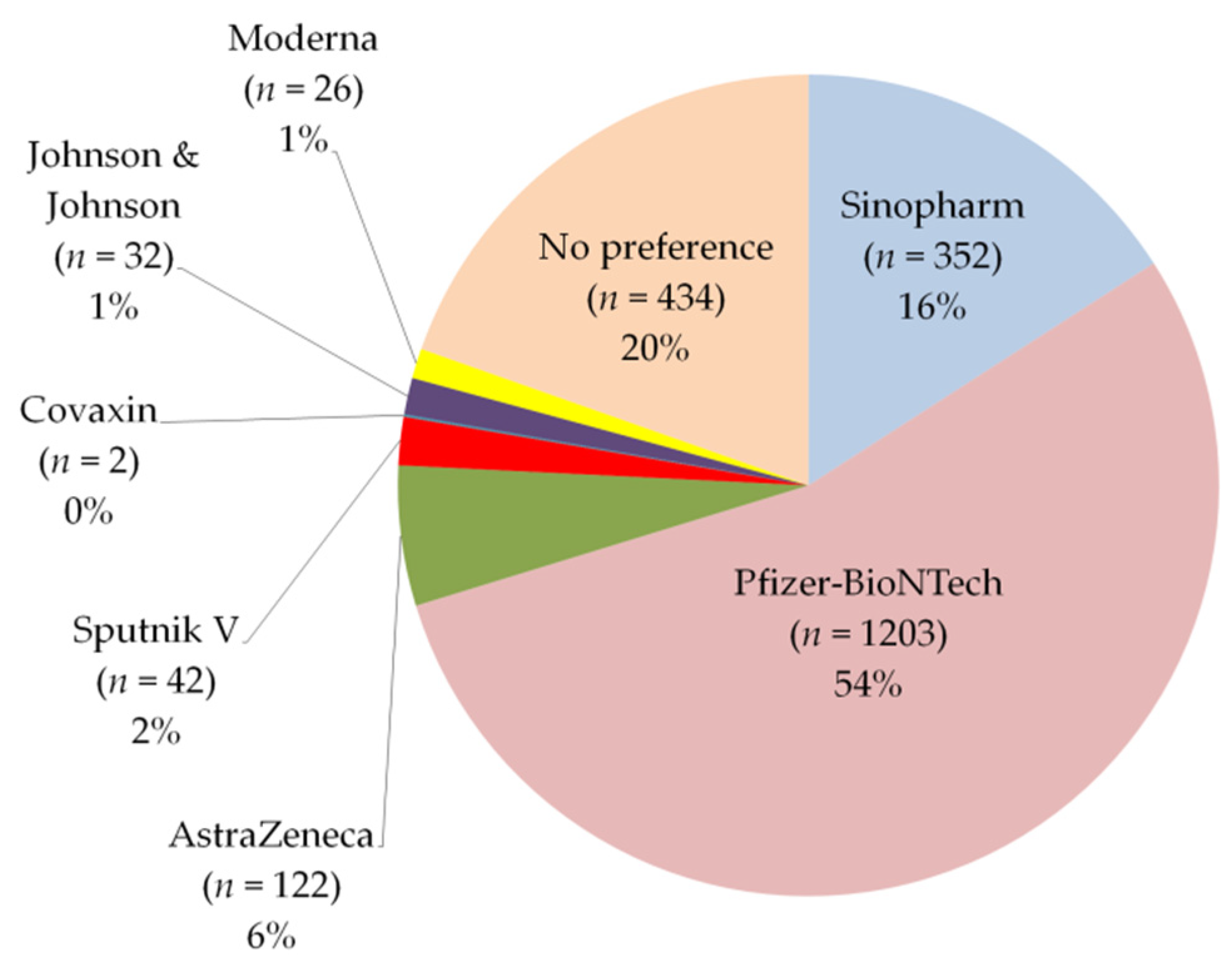
Vaccines | Free Full-Text | Side Effects and Perceptions Following COVID-19 Vaccination in Jordan: A Randomized, Cross-Sectional Study Implementing Machine Learning for Predicting Severity of Side Effects
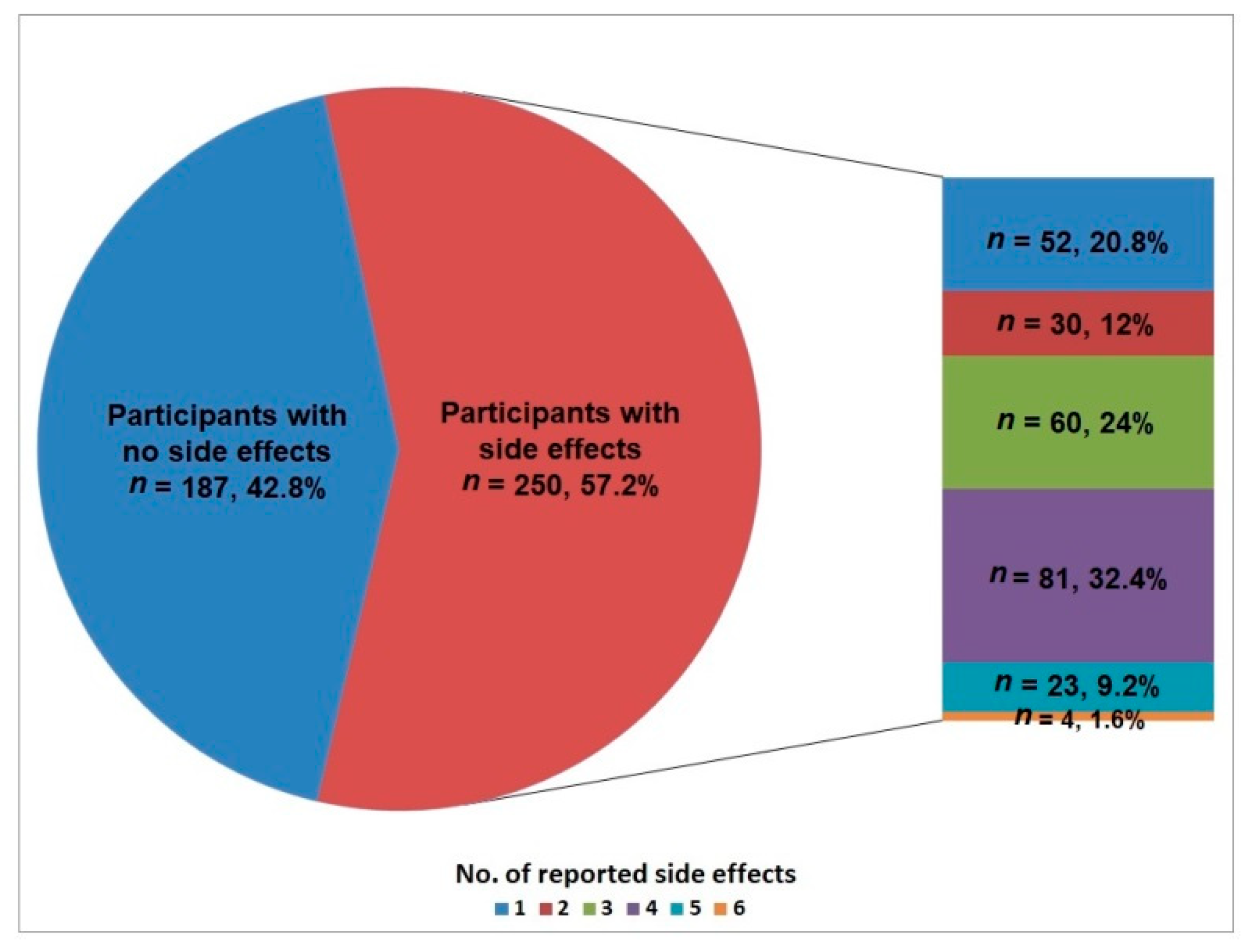
Vaccines | Free Full-Text | Prospective Evaluation of Side-Effects Following the First Dose of Oxford/AstraZeneca COVID-19 Vaccine among Healthcare Workers in Saudi Arabia

Study: the third dose of Comirnaty prevents death, severe disease and hospitalization – vaktsineeri.ee

COVID-19 Vaccine Boosters vs. Third Doses: Frequently Asked Questions - Anne Arundel County Department of Health

Interim Estimates of Vaccine Effectiveness of BNT162b2 and mRNA-1273 COVID-19 Vaccines in Preventing SARS-CoV-2 Infection Among Health Care Personnel, First Responders, and Other Essential and Frontline Workers — Eight U.S. Locations, December

Vaccine side-effects and SARS-CoV-2 infection after vaccination in users of the COVID Symptom Study app in the UK: a prospective observational study - The Lancet Infectious Diseases
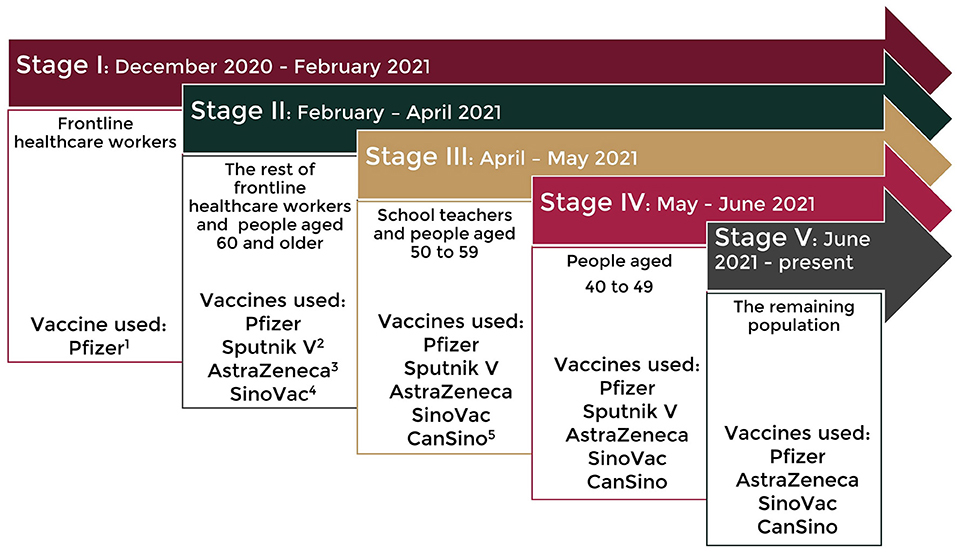
Frontiers | Extension and Severity of Self-Reported Side Effects of Seven COVID-19 Vaccines in Mexican Population

Comparison of medium-term adverse reactions induced by the first and second dose of mRNA BNT162b2 (Comirnaty, Pfizer-BioNTech) vaccine: a post-marketing Italian study conducted between 1 January and 28 February 2021 | European

/cloudfront-us-east-2.images.arcpublishing.com/reuters/2JFQTADGE5MYRP2IWXHXQ6JIBI.jpg)
/cloudfront-us-east-2.images.arcpublishing.com/reuters/ZOXP3JG4SVPBPAO7M46YGJVUU4.jpg)