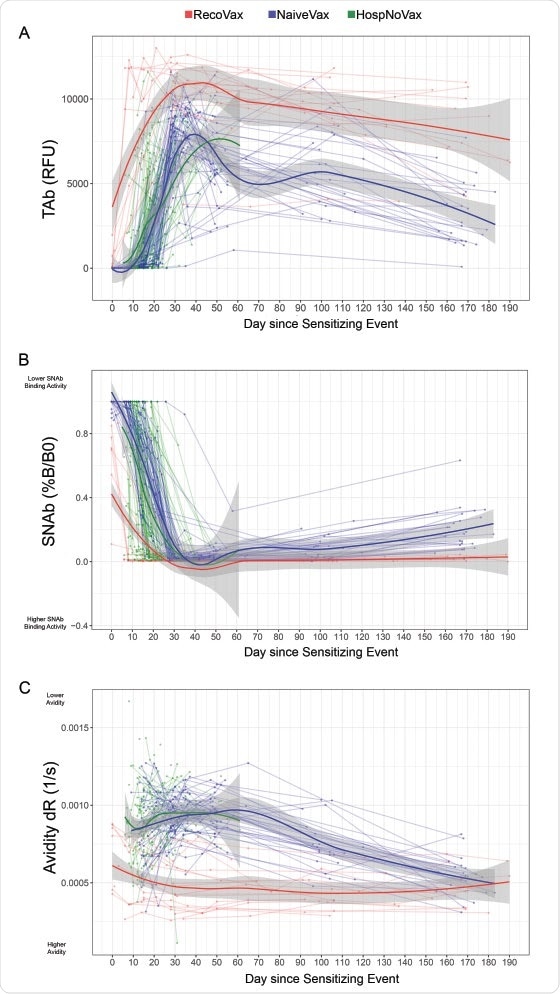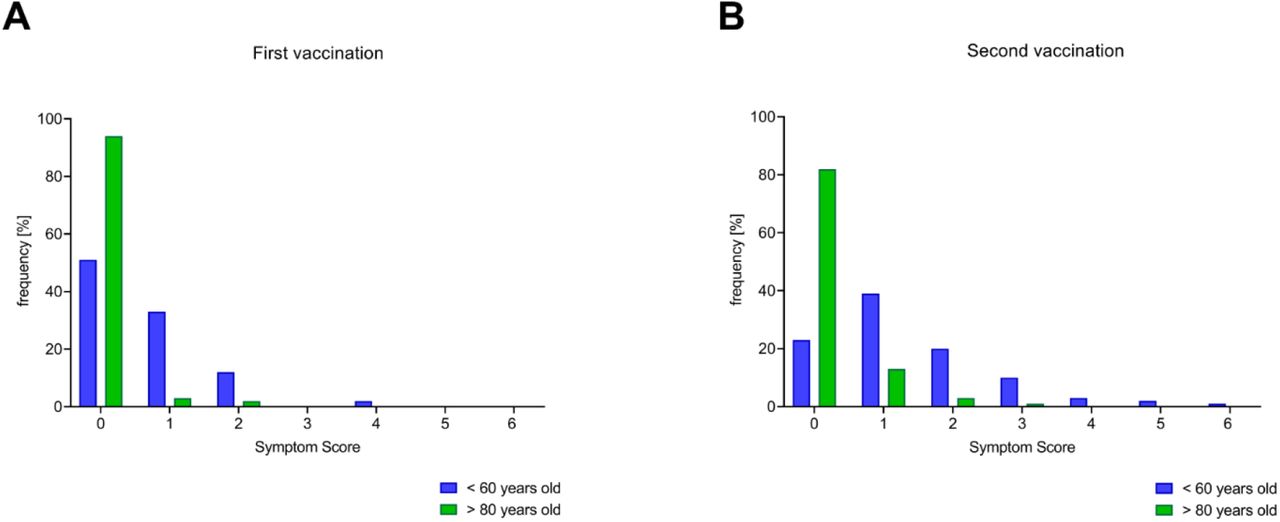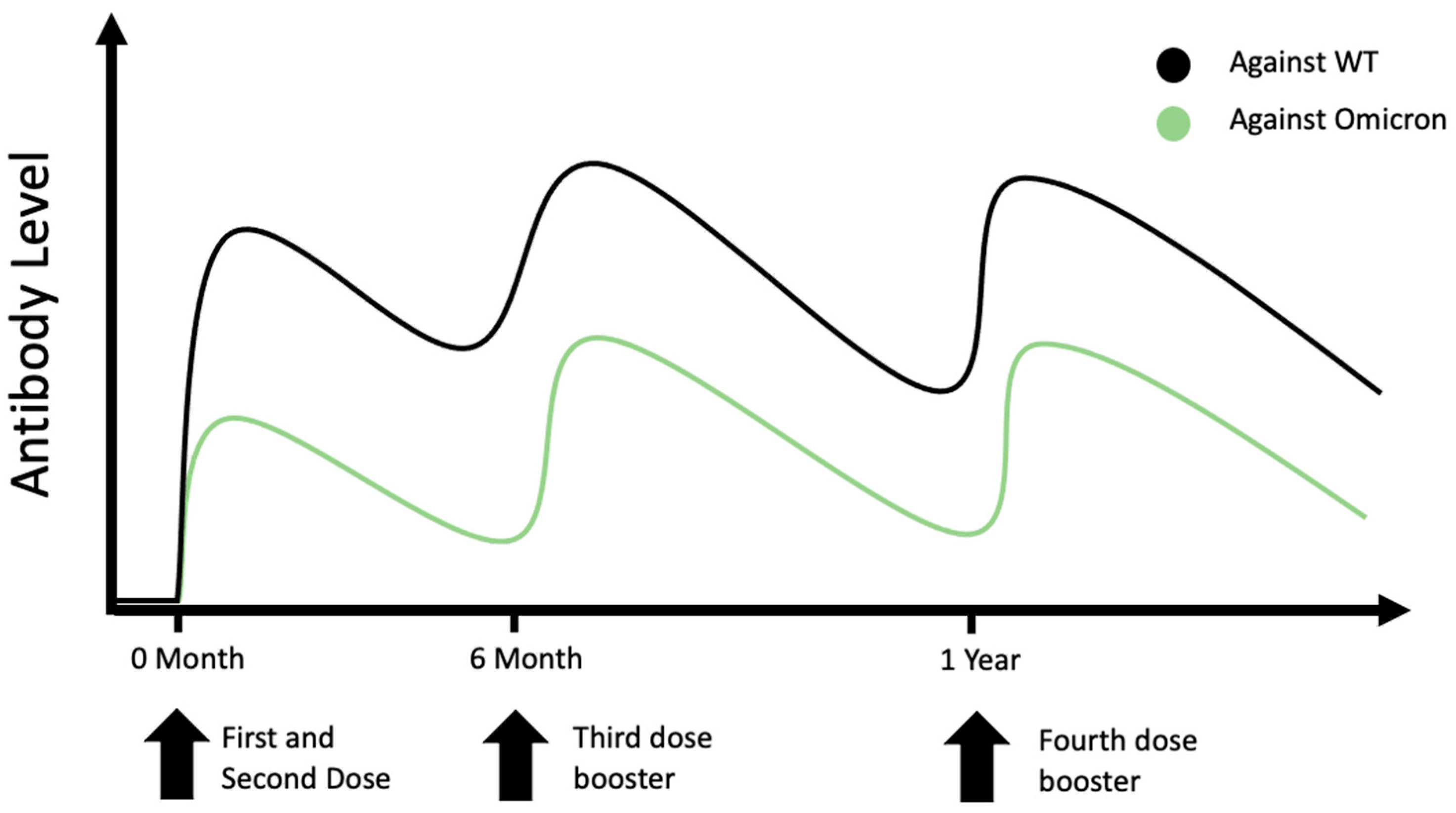
Vaccines | Free Full-Text | The Four Ws of the Fourth Dose COVID-19 Vaccines: Why, Who, When and What
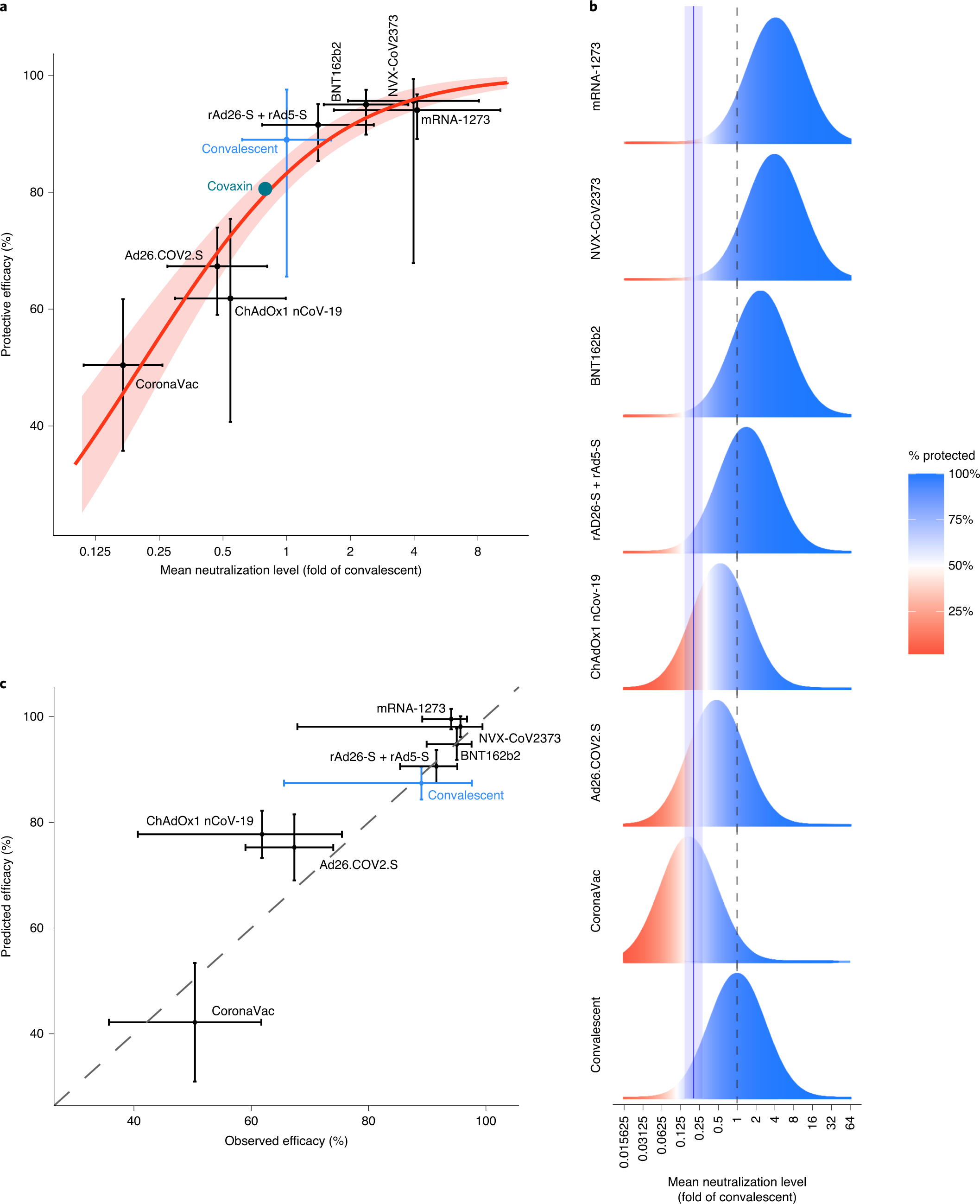
Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection | Nature Medicine
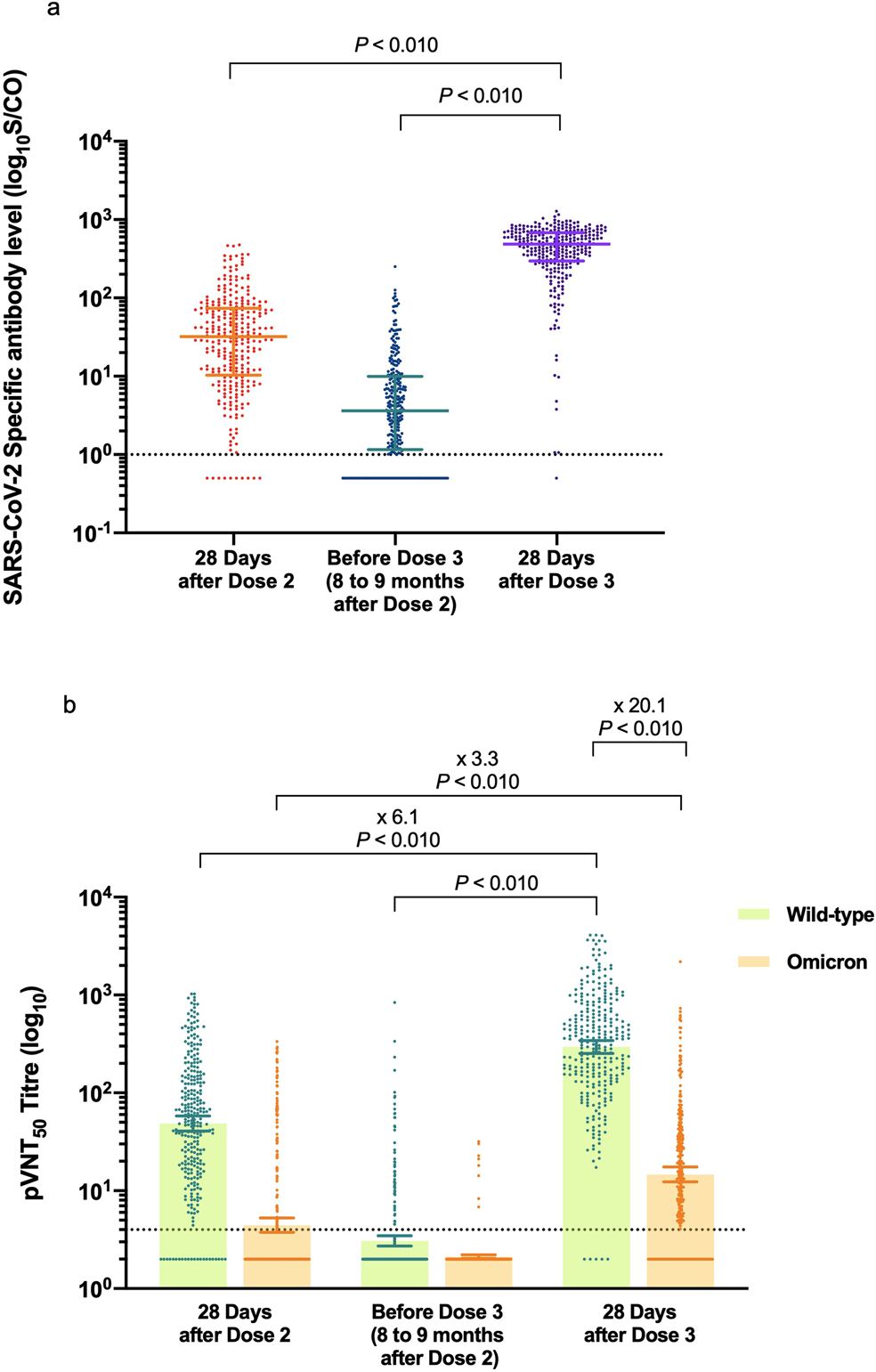
Reduced sensitivity of SARS-CoV-2 Omicron variant to antibody neutralization elicited by booster vaccination | Cell Discovery
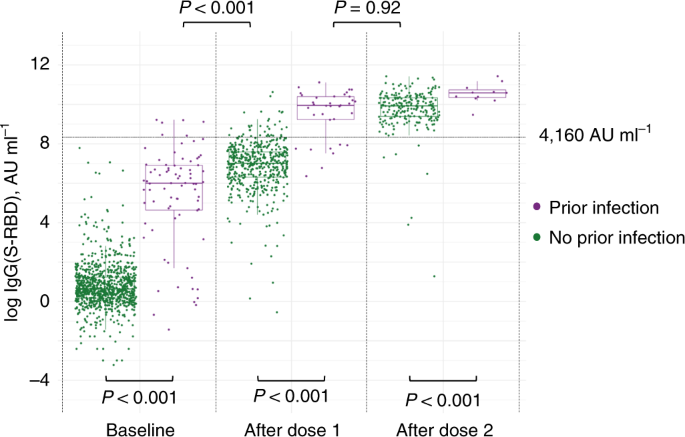
Antibody responses to the BNT162b2 mRNA vaccine in individuals previously infected with SARS-CoV-2 | Nature Medicine

Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study - The Lancet

Third COVID-19 vaccine dose with BNT162b2 in patients with ANCA-associated vasculitis | Annals of the Rheumatic Diseases
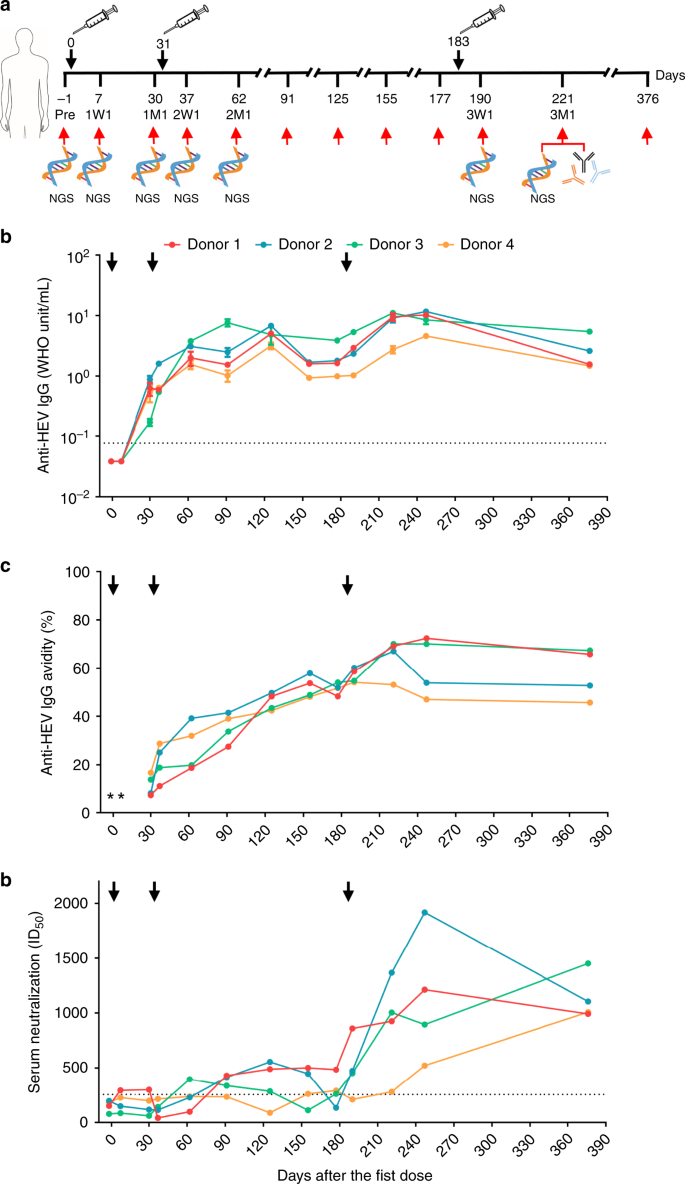
Quantitative evaluation of protective antibody response induced by hepatitis E vaccine in humans | Nature Communications

Vaccination before or after SARS-CoV-2 infection leads to robust humoral response and antibodies that effectively neutralize variants | Science Immunology
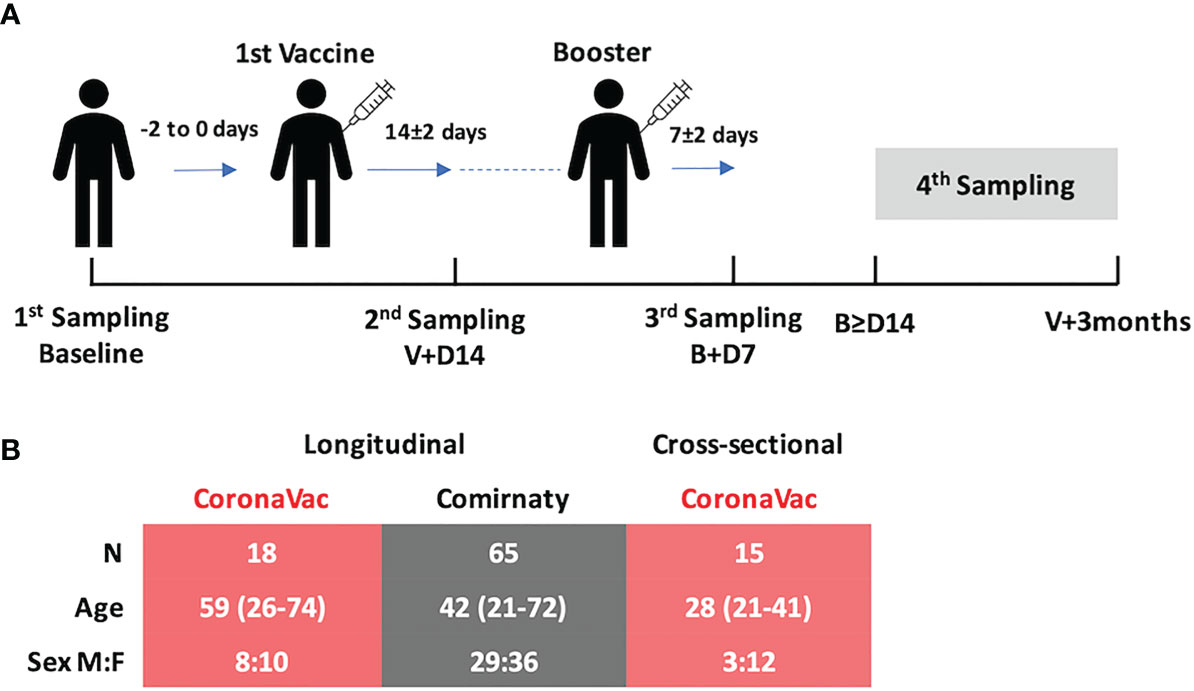
Frontiers | The Mucosal and Serological Immune Responses to the Novel Coronavirus (SARS-CoV-2) Vaccines
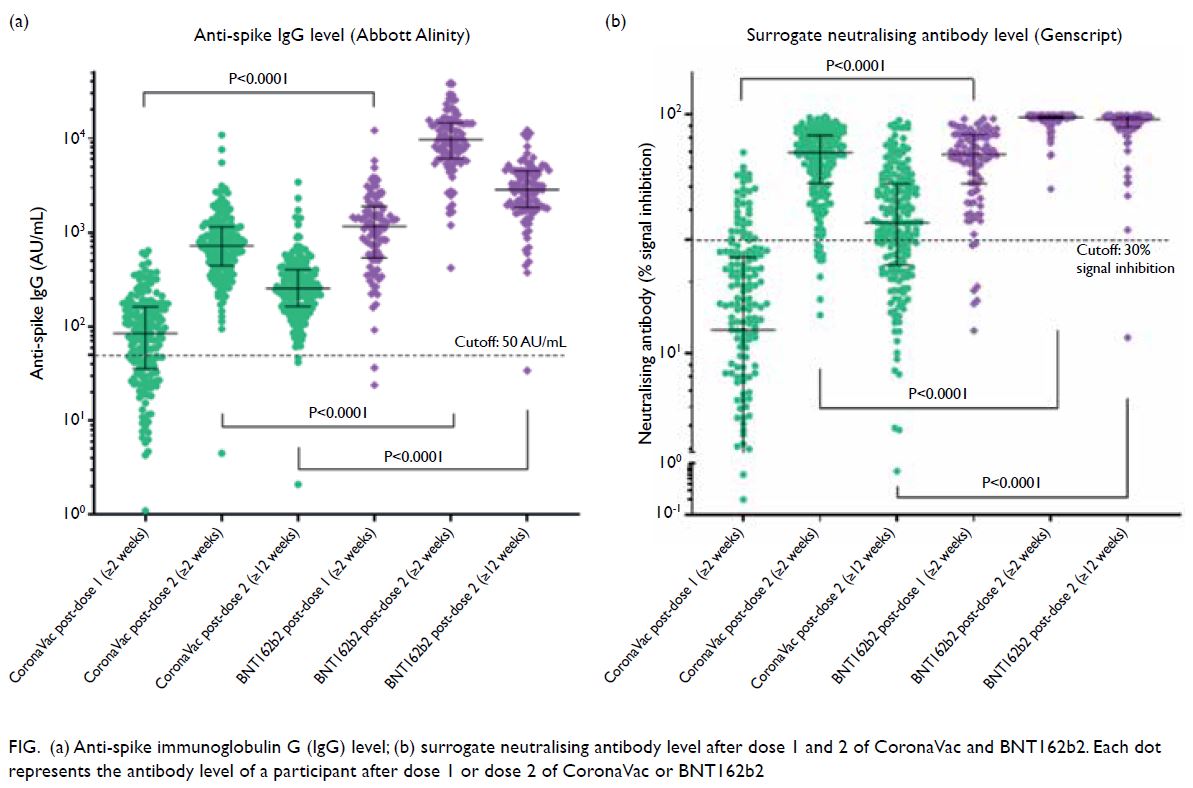
Serological response to mRNA and inactivated COVID-19 vaccine in healthcare workers in Hong Kong: decline in antibodies 12 weeks after two doses | HKMJ
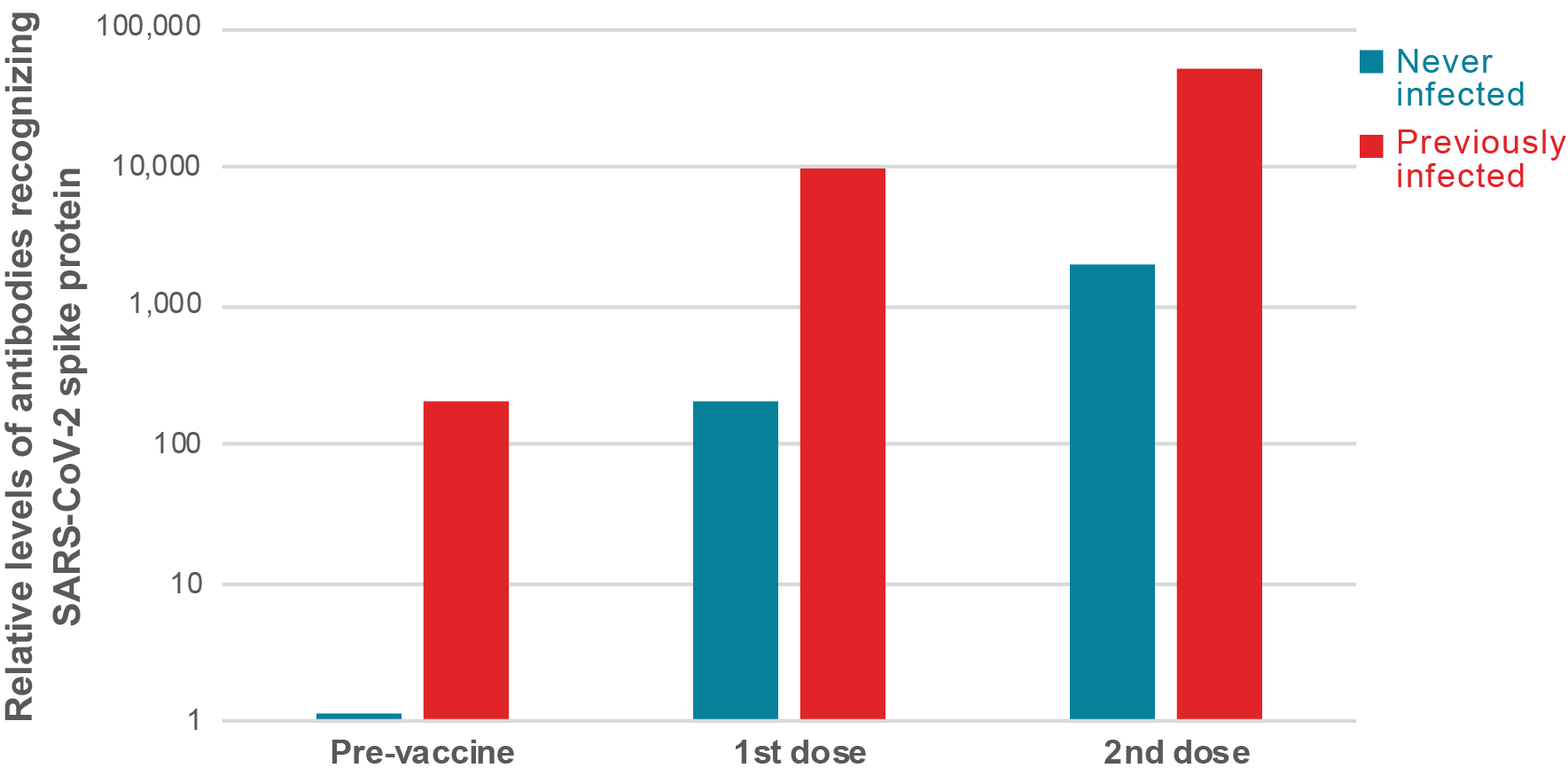
Several studies suggest single dose of vaccine provides strong protection for people who have recovered from COVID-19 - COVID-19 Immunity Task Force

Comparison of IgG and neutralizing antibody responses after one or two doses of COVID-19 mRNA vaccine in previously infected and uninfected individuals. - eClinicalMedicine

Comparison of SARS-CoV-2 anti-spike receptor binding domain IgG antibody responses after CoronaVac, BNT162b2, ChAdOx1 COVID-19 vaccines, and a single booster dose: a prospective, longitudinal population-based study - The Lancet Microbe
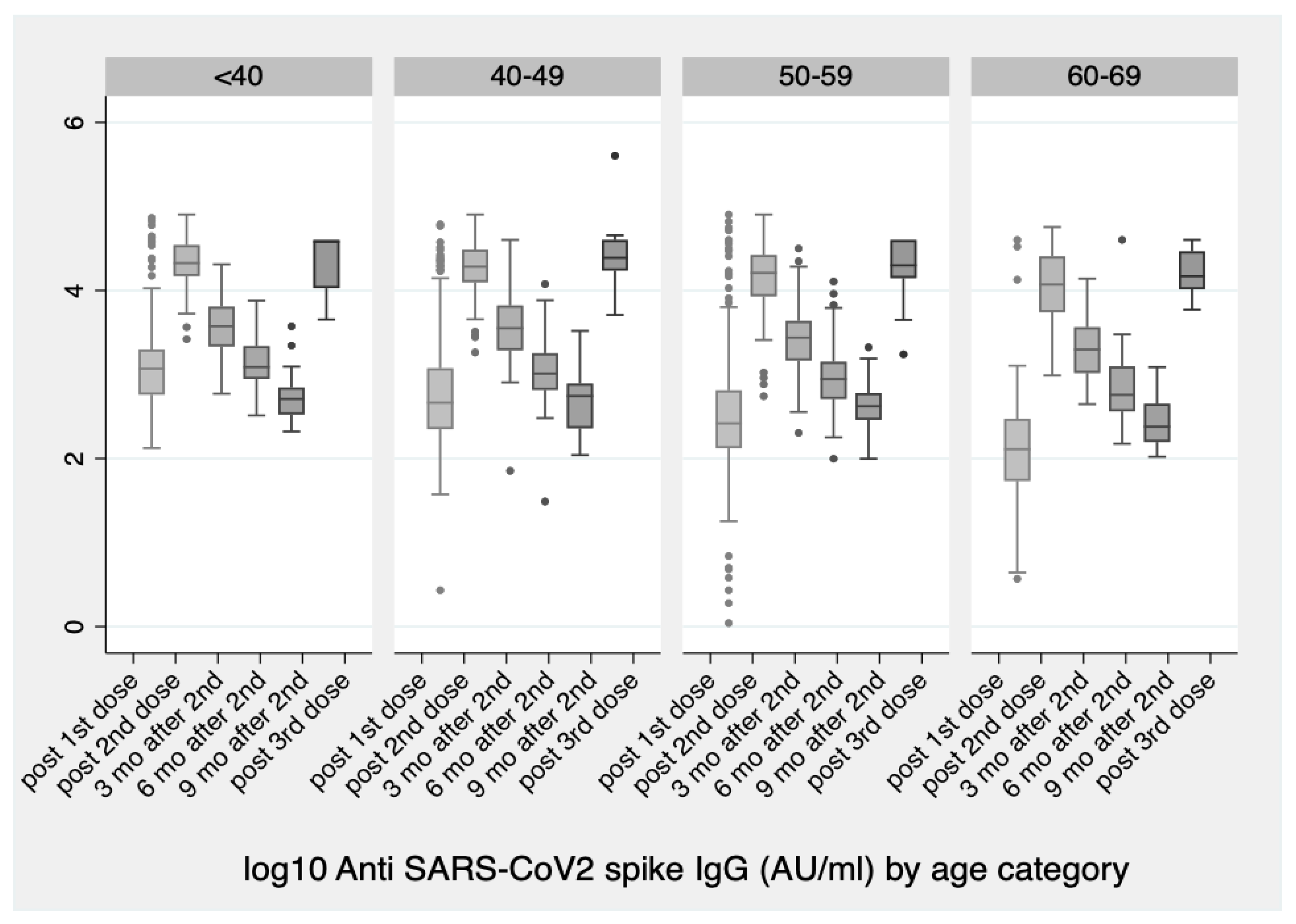
Vaccines | Free Full-Text | Significant Increase in Antibody Titers after the 3rd Booster Dose of the Pfizer–BioNTech mRNA COVID-19 Vaccine in Healthcare Workers in Greece